[Published] 2025/3/25 [Last updated] 2025/3/25
Table of Contents
Short summary: New drug development in JAPAN
- Japan as a Leading Pharmaceutical Market
- All drugs approved in Japan are simultaneously covered by the national health insurance system, ensuring broad patient access.
- Next-generation sequencing (NGS) genetic testing is also included in the national health insurance coverage, promoting advanced precision medicine.
- The Pharmaceuticals and Medical Devices Agency (PMDA) has basically accepted global clinical trial data, facilitating international drug development.
- Conducting clinical trials in Japan provides multiple advantages, including: (PoC establishment, Translational, Expansion into the broader Asian market)
- Hospitalization during the dose-limiting toxicity (DLT) evaluation period is not uniformly required, providing flexibility in trial design and patient management.
Japan is 3rd drug market in globally
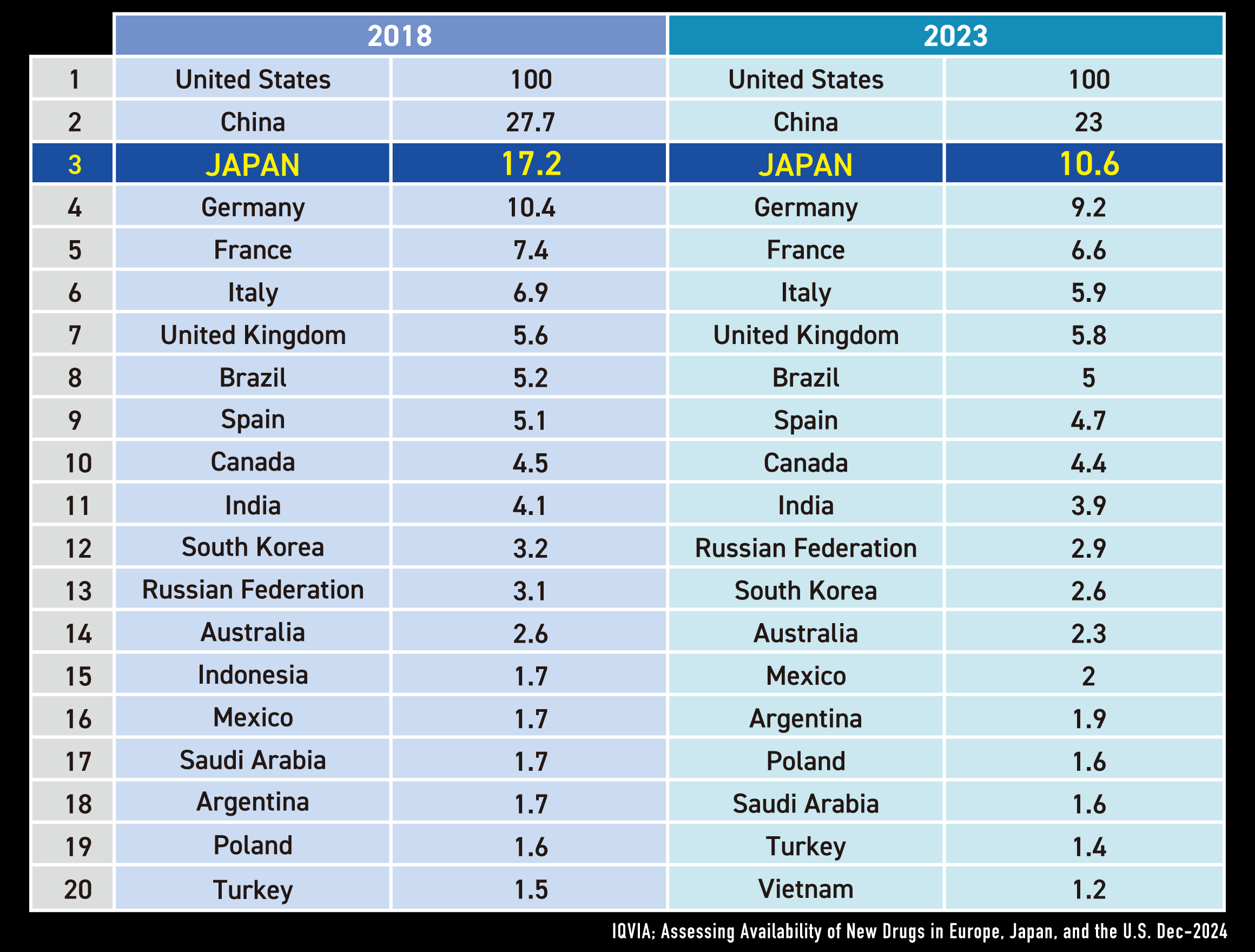
Japan Snapshot

Feasibility / Possibility of Japan involvement
Japan’s involvement is feasible
- PMDA has the capability to accept global clinical trial data, facilitating the inclusion of Japan in international drug development initiatives.
- NCCH could actively contribute to patient enrollment, enhancing the feasibility of clinical trials in Japan.
Japanese branch is not required
- care-takers (ICCC), such as CROs, are fully capable of managing clinical trials in Japan, eliminating the necessity for a local corporate entity.
- ICCC can effectively manage regulatory submissions, including CTN and IND applications, ensuring regulatory compliance.
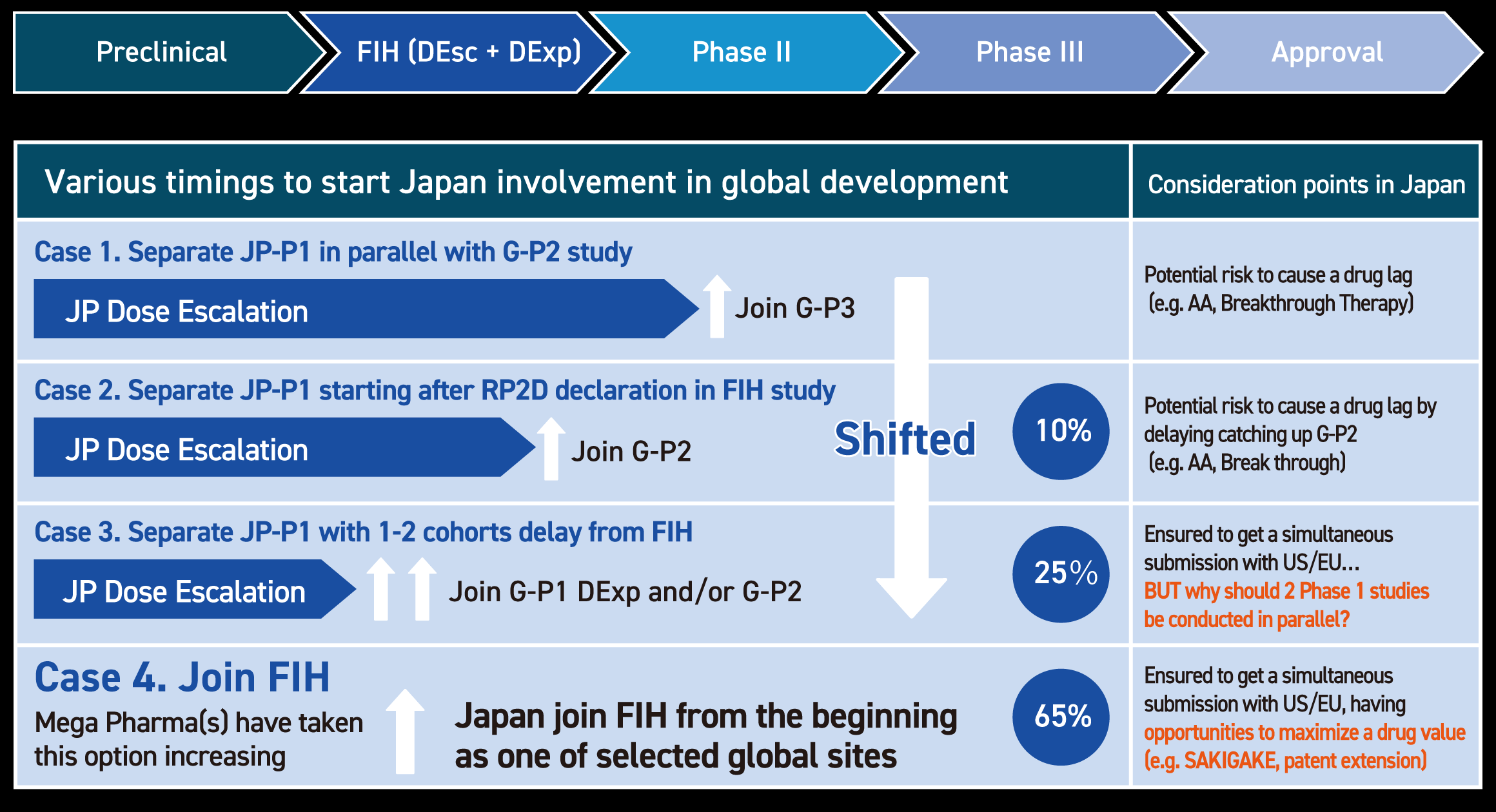
Ideal approach in new drug development in Japan
1st recommendation
- Inclusion of Japanese Clinical Sites in Early Drug Development
- Conducting First-in-Human (FIH) and/or Phase I Trials in Japan
2nd recommendation (after FIH and phase I trial in US)
- Prior to the pivotal clinical trial, a feasibility trial involving a small number of Japanese patients can be conducted to assess the applicability and effectiveness of the investigational treatment in the Japanese population.
- A safety run-in cohort may also be acceptable, depending on the study timeline, to ensure early safety assessment and optimal dose selection.
Advantage of Japan involvement
Expanding the Development of Innovative Therapeutics
- Facilitated POC evaluation through High-Performance Serial Tumor Biopsy
- The high-quality human samples (tumor tissues) and AI reserches at NCC facilitates seamless collaboration in TR/rTR.
- Conducting research in Japan enables the identification of critical biomarkers and signal findings in Asian-prevalent cancers, such as gastric cancer, cholangiocarcinoma, and EGFR-mutant non-small cell lung cancer (NSCLC).
All approved medicines in Japan are simultaneously covered by the national health insurance system
- Once a drug receives regulatory approval, it is immediately covered by the national health insurance system, ensuring broad patient access and predictable market entry.
Expansion of New Drug Development Opportunities
- Facilitated POC Evaluation through Serial Tumor Biopsy
- Seamless Collaboration in Translational and Reverse Translational Research (TR/rTR)
- Identification of Key Biomarkers in Asian-Specific Cancers
Paradigm shift of Japan involvement timing, Current