[Published] 2025/3/4 [Last updated] 2025/3/25
Table of Contents
Short summary : Achievements of new drug development
Recent year in review of our phase 1 program, the total number of patients enrolled into trials has been exceeded more than 270 and the total number of both ongoing phase 1 trials and the newly opened trials are increasing as figure is indicating.
- 101
- Number of ongoing
Phase 1 trials
- 270
- Number of Patients per year
enrolled in phase 1 trials
- 39
- Number of ongoing FIH
phase 1 trials
Industry Sponsored trials (ISTs) and Investigator Initiated Trials (IITs) in NCCH
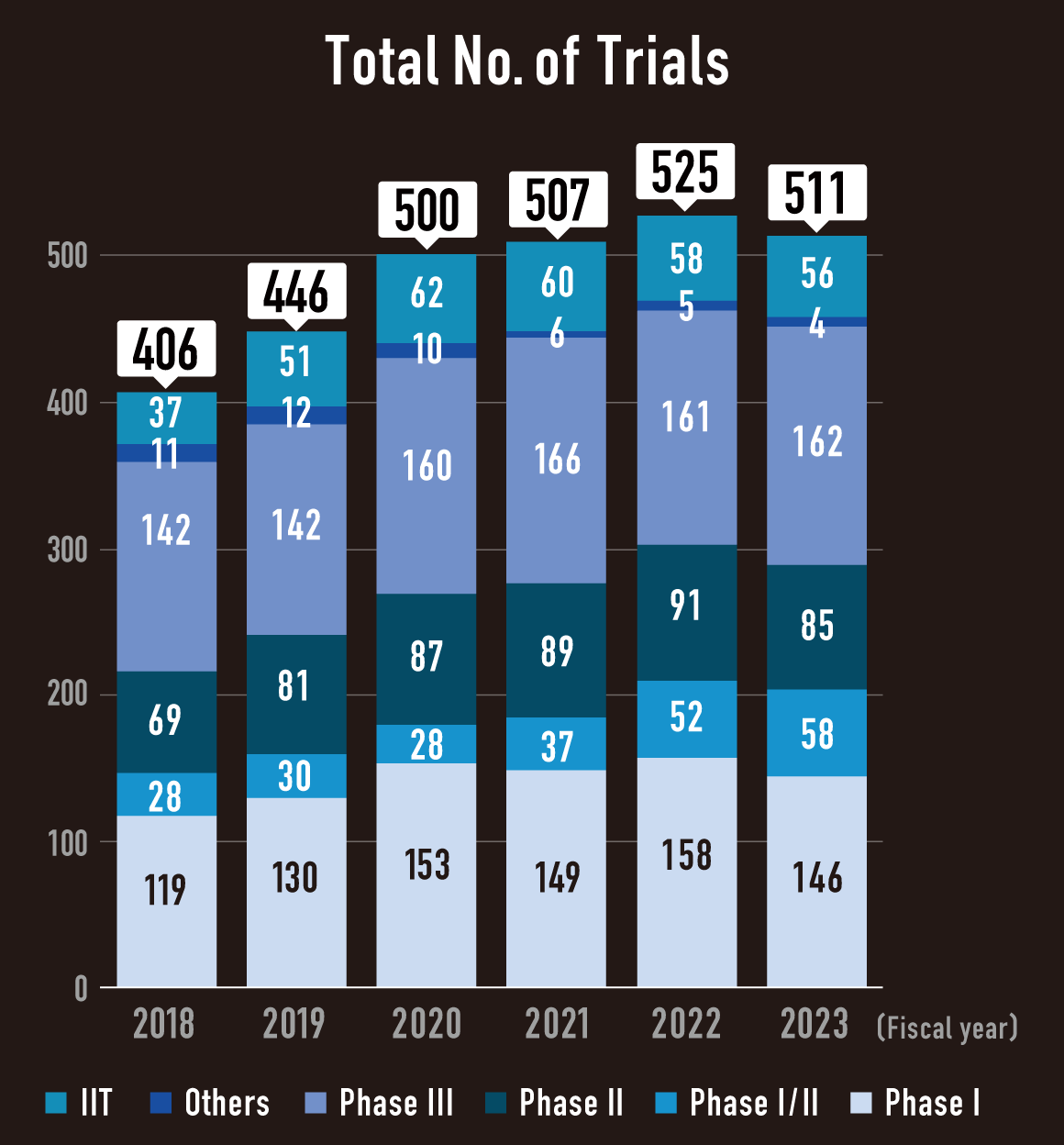
- 90% of trials are industry sponsored trials.
- 60% of trials are global trial.
- High priority for phase I and FIH trials
- IIT is mainly focused on rare cancer and pediatric malignancies.
Why FIH and phase I trials?
Recent new drug development is……
- Globalized and Individualized
- Target indication is highly focused on genetic alterations
- Pivotal trial is not always large scale
Single arm phase II trial is implemented as a pivotal trial
If we could participate FIH or phase I trials……
- Feasibility could be evaluated in minimum number of Japanese.
- Maintain drug development speed in Japan (US and Japan)
- Achieve drug approval in the same time (US and Japan)
Other options
- Propose new development (i.e., other indication) during FIH experience
- Propose TR to explore POC, resistance mechanism, etc.
FIH trials in NCCH
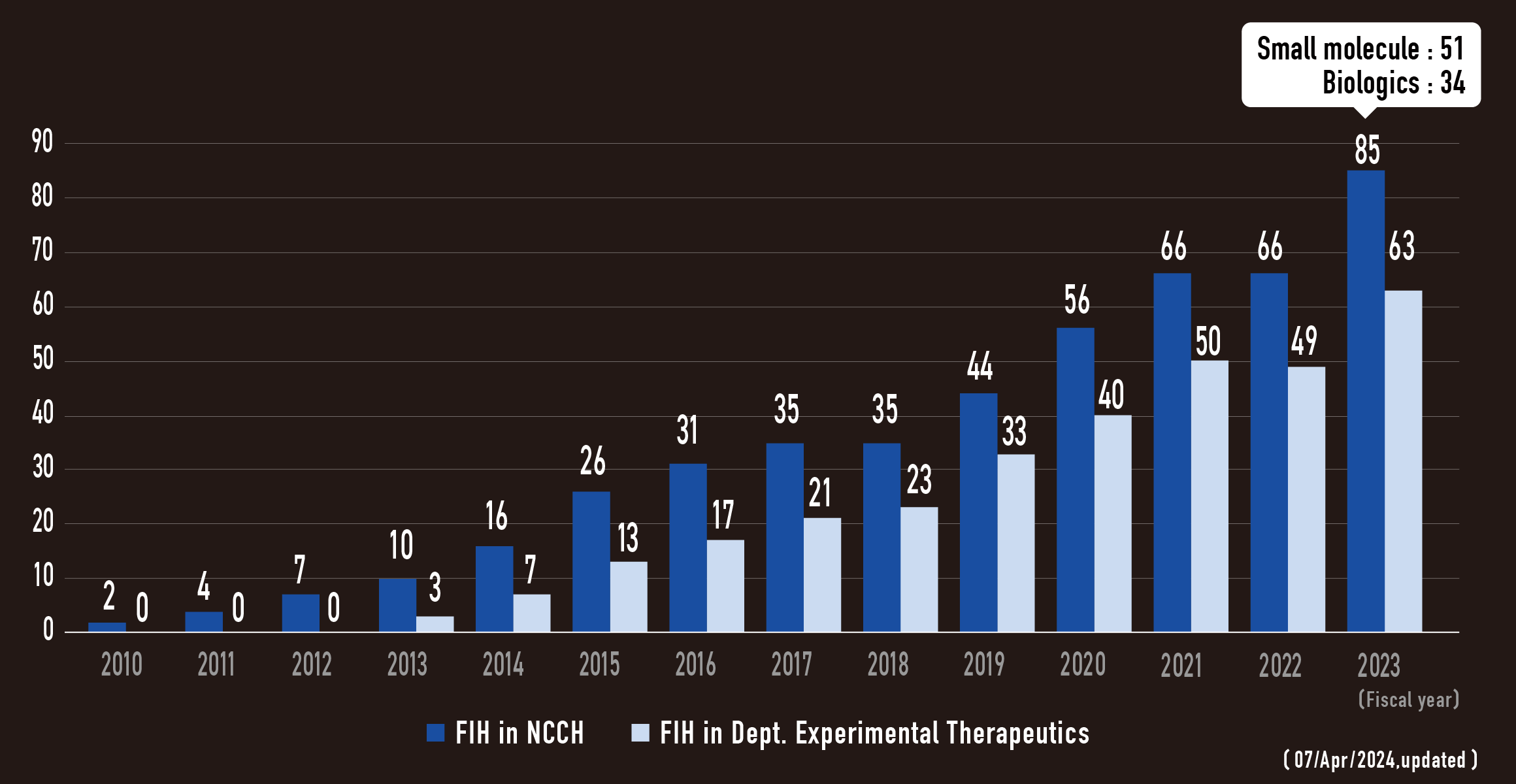
Phase I trials in Dept. Experimental Therapeutics
FIH trials in Dept. Experimental Therapeutics
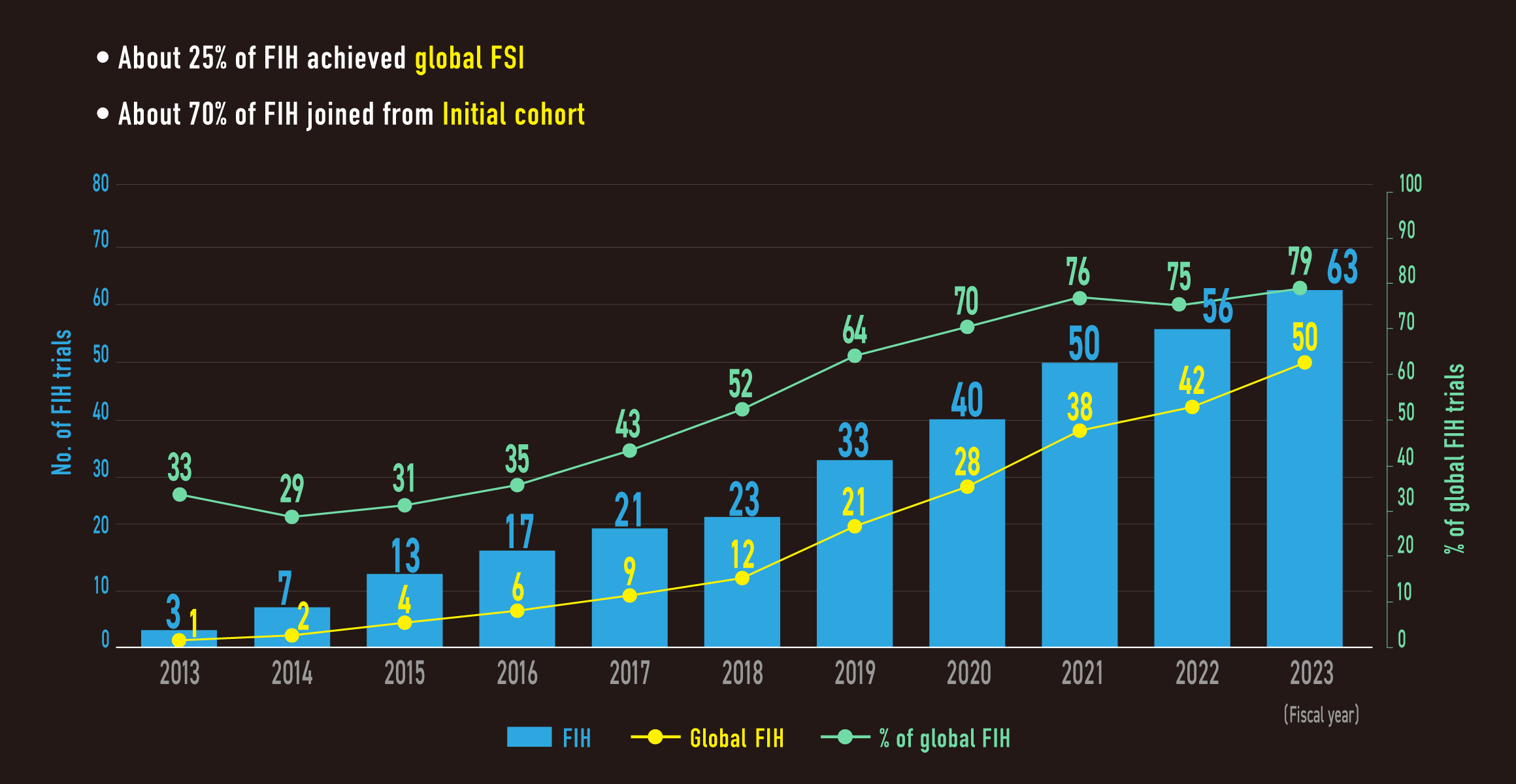
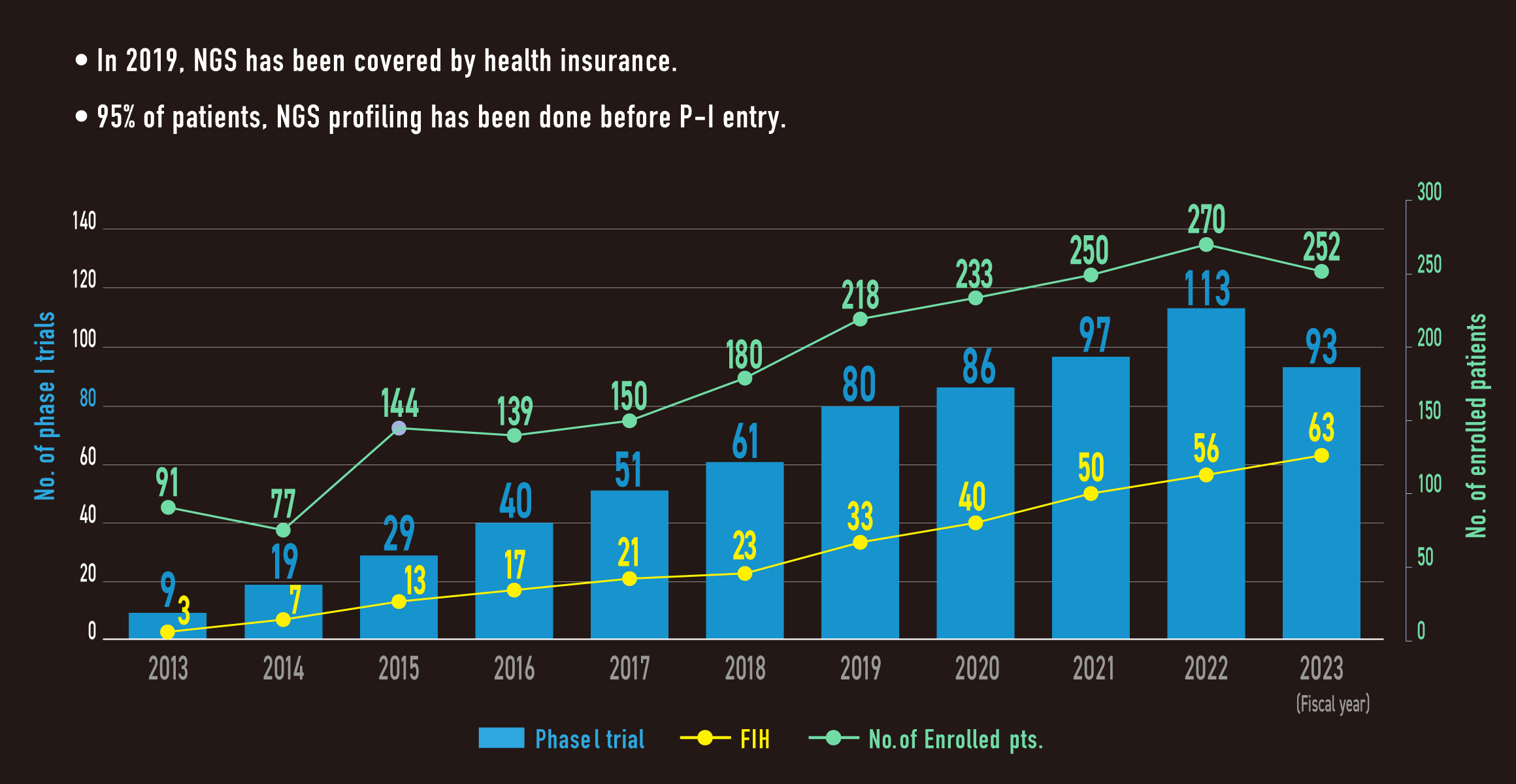
Key to achieve global FSI and initial cohort commitment
In NCCH
- Recruit performance
- High frequency (95%) of NGS screening prior to visit phase I clinic
- Rapid preparation from IRB submission to contract
Ave. 29.5 days in FY2023
Outstanding performance in pharma Japan-team
- Abbvie, Novartis, Boehringer, Eli Lilly, etc.
Annual change in the number of cases performed NGS
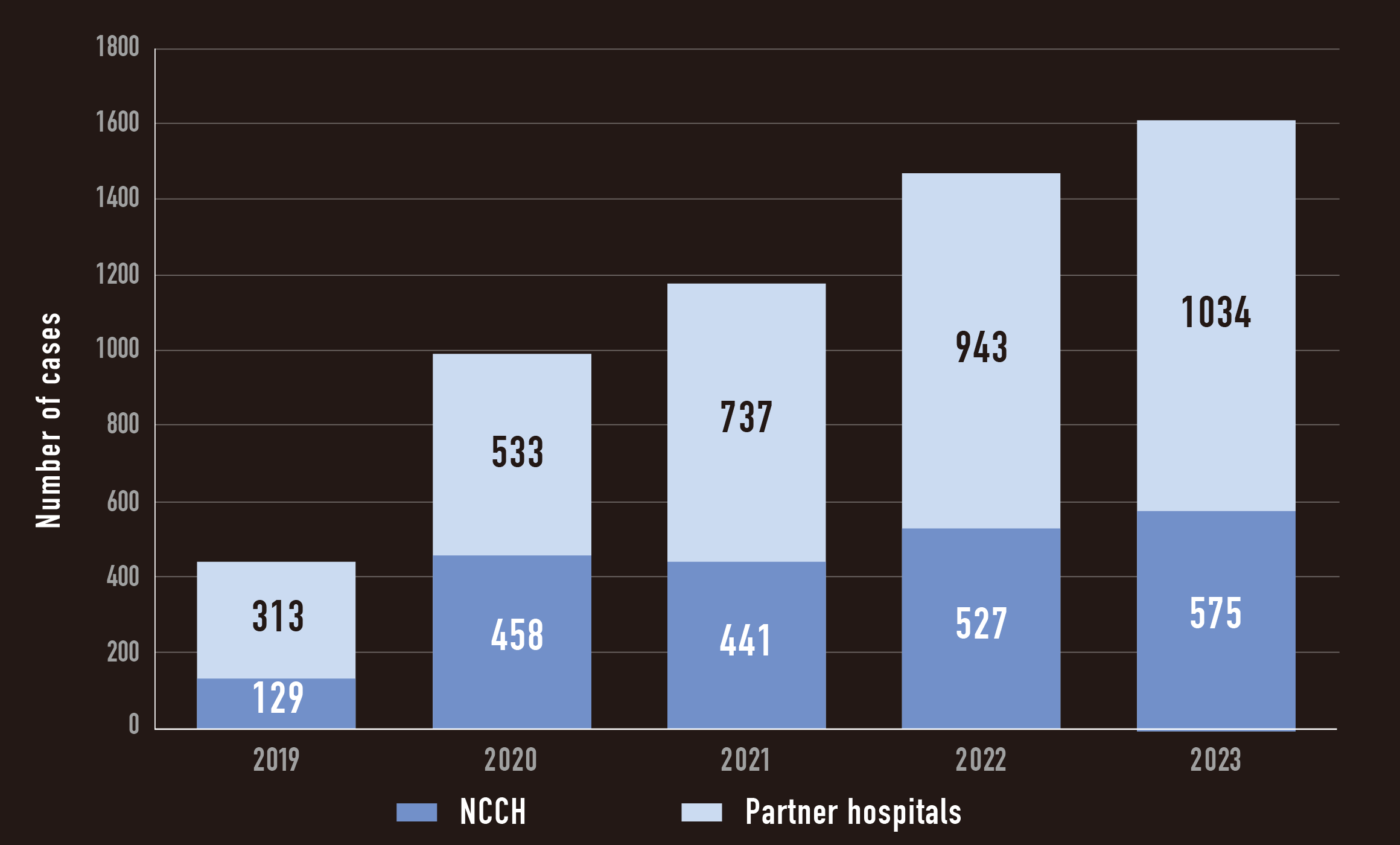
Advantage of Japan involvement, Business aspect
Rapid preparation (site open)
- Ave. 29.8 days in FY2023
From IRB submission (& review) to IRB:26.6 days
From IRB to contract:3.2 days
Low price per case
- FIH, iv agent (ADC), q3wks, full-pk sampling, serial tumor biopsy, 4 cycles: 22,000 USD/case
- FIH, oral agent, 3wks/cycle, full-pk sampling, serial tumor biopsy, 4 cycles: 25,000 USD/case
Hospitalization
- Hospitalization fee is paid by patient in Japan
- From 70 to 90% of hospitalization fee is covered by health insurance in Japan
Experience FIH from biotech
| Drug | Type | Tumor | Phase | Sponsor | Status | Trial No. |
|---|---|---|---|---|---|---|
| NIB101 | CAR-T | GM2 solid tumor | I | Noile-Immune Biotech, Inc | Recruiting | NCT05192174 |
| CTX-712 | CLK inhibitor | Solid tumor | I | Chordia Therapeutics, Inc. | Recruiting | jRCT2080224127 |
| CBA-1205 | Anti DLK-1 antibody | Solid tumor | I/Ib | Chiome Bioscience Inc. | Recruiting | jRCT2080225288 |
| CBA-1535 | Tribody | Solid tumor | I | Chiome Bioscience Inc. | Recruiting | jRCT2031210708 |
| AS-0141 | CDC7 inhibitor | Solid tumor | I | Carna Biosciences, Inc. | Recruiting | jRCT2031210072 |
Another commitment in phase I and FIH
New additional development proposal

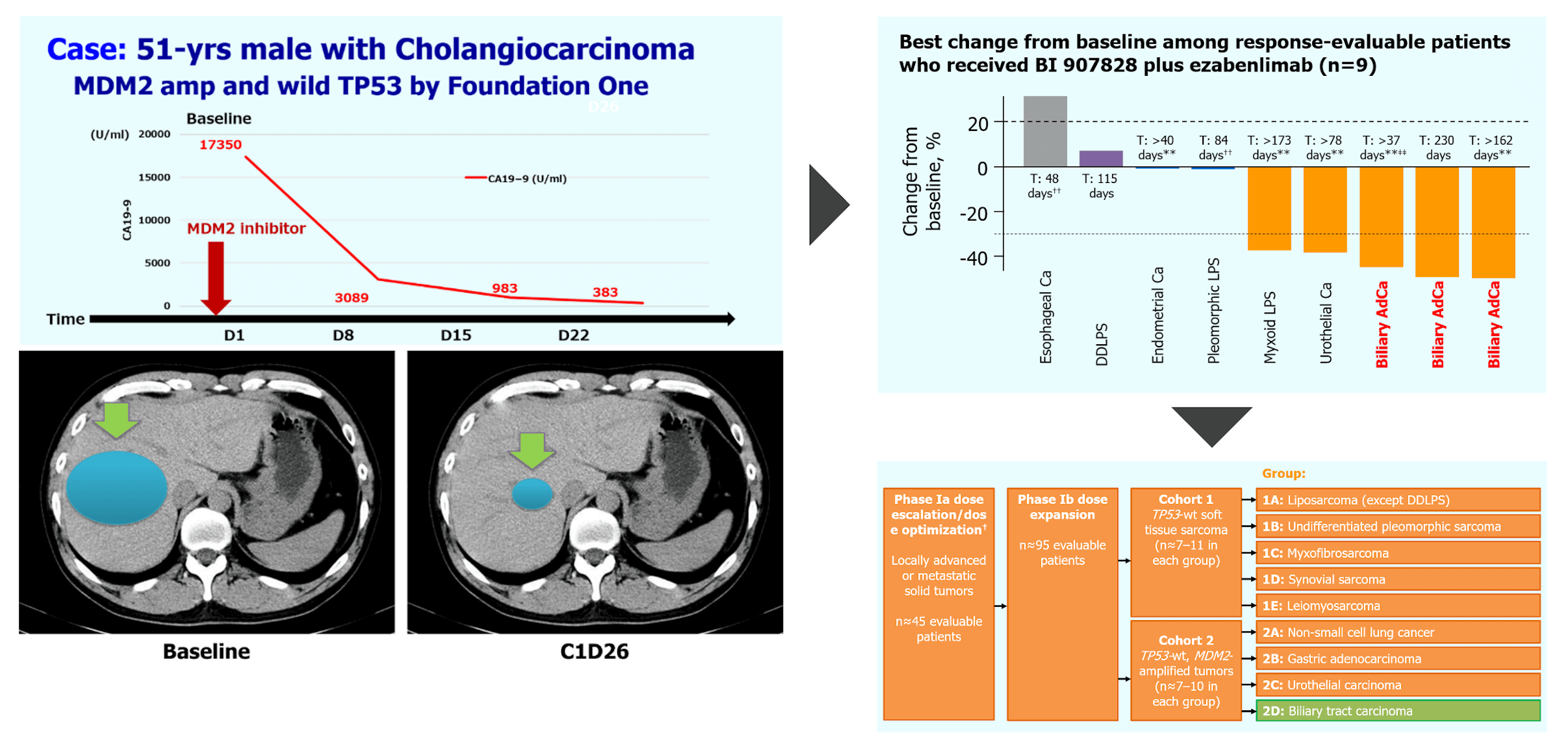
(ASCO 2022, #3095)